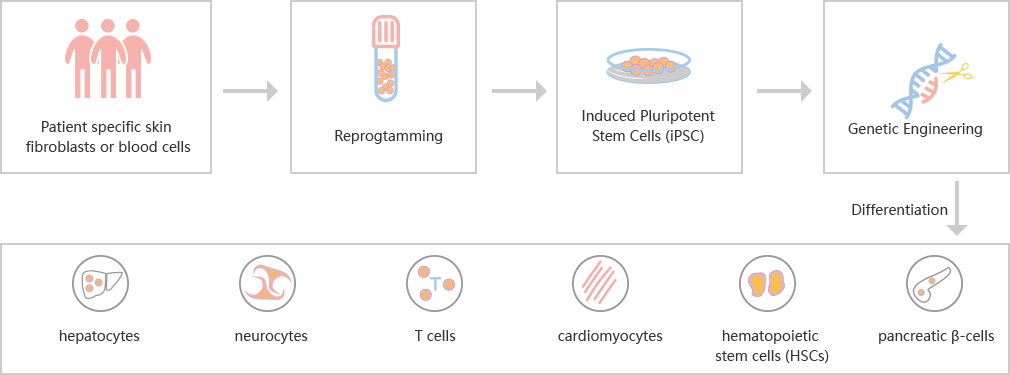
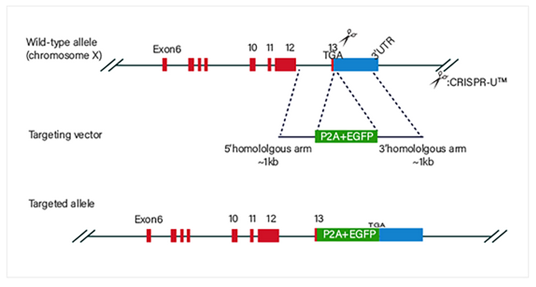
CRISPR-U™ gene knockout iPSC cell line: gRNA and Cas9 are
transferred
into iPSCs by nucleofection. After drug screening, single clones would
be
generated. Positive clones would be validated by sequencing.
|
Type
|
Strategy
|
Application
|
|
Short fragment removal
|
Guide RNAs target introns at both sides of exon 2 and the number of bases in exon 2 is not a multiple of 3, which can cause frame-shift mutation.
|
Study of gene function through gene defect
|
|
Frame-shift mutation
|
Guide RNA targets the exon, and the base number of deletion is not a multiple of 3. After knockout, frame-shift mutation would cause gene knockout.
|
|
Large fragment removal
|
Complete removal of the coding sequence to achieve gene knockout.
|
Case Study:
The limited T cells and the difficulty of
proliferation is
the main obstacle of T-cell immunotherapy, which can be overcome by
using
pluripotent stem cells with proliferation and differentiation ability to
generate T-iPSC with antigen specificity. Strict antigen specificity is
essential for safe and effective T-cell immunotherapy. However, in the
process of double-positive CD4/CD8 differentiation, the rearrangement of
the
T-cell receptor (TCR) α chain will lose antigen specificity. This TCR
rearrangement was prevented by removing the recombinant enzyme gene
(RAG2)
in T-iPSCs with CRISPR/Cas9. Xenotransplantation of CD8αβ-T cells with
stable TCR can effectively inhibit tumor growth in disease models. This
contributes to a safe and effective T-cell immunotherapy.
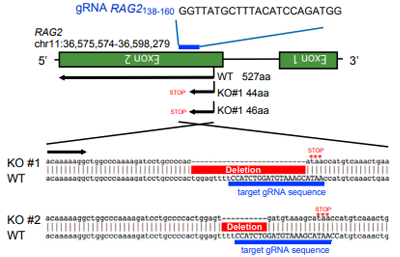
gRNA sequence and RAG2ockout sequence。The positive
clones
have frameshift mutations in the designated RAG2.
Comparison of the binding ability of WT and RAG2
knockout
T-iPSCs to dextramer. RAG2-/- T-iPSCs differentiated into CD8αβ cells
expressing stable TCR, while 40% of RAG2wt/wt-iPSC derived CD8αβ cells
lost
antigen specificity.
Reference:
Minagawa, Atsutaka, et al. "Enhancing T cell receptor stability in
rejuvenated iPSC-derived T cells improves their use in cancer
immunotherapy." Cell Stem Cell 23.6 (2018): 850-858.
Point Mutation
iPSC would be co-transfected with gRNA, Cas9 and donor oligo by
electroporation. After the DNA DSB caused by the complex of gRNA and
Cas9,
iPSCs use donor oligo carrying wild-type sequence as a template for
homologous recombination repair (HDR) and replace the target sequence
with
point mutation.
Case Study:
 Disease model generation
Disease model generation
ssODN carrying point mutation which replaces the WT sequence by HDR.
 Disease model rescuing
Disease model rescuing
ssODN carrying WT sequence which replaces the mutated site by HDR.
The limited T cells and the difficulty of
proliferation is
the main obstacle of T-cell immunotherapy, which can be overcome by
using
pluripotent stem cells with proliferation and differentiation ability to
generate T-iPSC with antigen specificity. Strict antigen specificity is
essential for safe and effective T-cell immunotherapy. However, in the
process of double-positive CD4/CD8 differentiation, the rearrangement of
the
T-cell receptor (TCR) α chain will lose antigen specificity. This TCR
rearrangement was prevented by removing the recombinant enzyme gene
(RAG2)
in T-iPSCs with CRISPR/Cas9. Xenotransplantation of CD8αβ-T cells with
stable TCR can effectively inhibit tumor growth in disease models. This
contributes to a safe and effective T-cell immunotherapy.

CRISPR/Cas9 and ssODN used to repair the point
mutation in
A79V-hiPSC. A) Genomic sequence surrounding the mutation site: mutated
nucleotide (T, red); sgRNA recognition site containing 20 bp (yellow);
CRISPR cutting site between the 17th and 18th bp (bold); forward and
reverse
primers (pink). B) ssODN with 120 bp, 60 bp upstream and 60 bp
downstream
the mutation site containing the WT nucleotide (C, green).
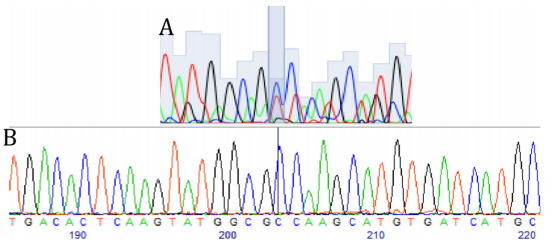
Sequencing of exon 4 of the PSEN1 gene in hiPSCs.
A)
Heterozygous c.236C>T substitution in the mother line previously
published.
B) Successful correction of the point mutation (T>C).
Reference:
Pires, C., Schmid, B., Petræus, C., Poon, A., Nimsanor, N., Nielsen, T.
T.,
... & Freude, K. K. (2016). Generation of a gene-corrected isogenic
control
cell line from an Alzheimer's disease patient iPSC line carrying a A79V
mutation in PSEN1. Stem cell research, 17(2), 285-288.
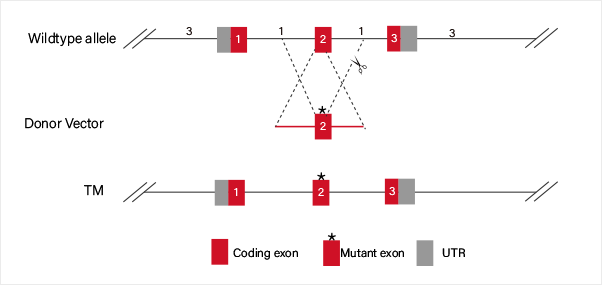
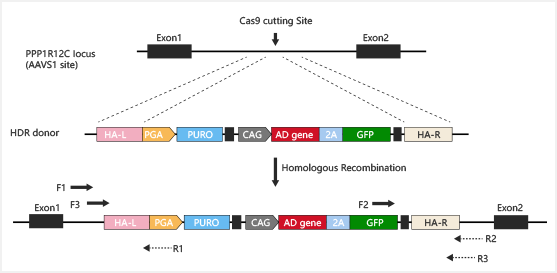
Knock in
CRISPR-U™ Gene Knockin iPSC:iPSC would be co-transfected with
gRNA, Cas9 and donor vector by electroporation. After drug
screening,
single clones would be generated. Positive clones would be validated
by
sequencing.
Knockin Strategies :
Disease model generation
Guide RNA and Cas9 complex cause a double-strand break (DSB) on the
target site of DNA. The donor vector carrying knockin sequence is
the
template for homologous recombination repair (HDR), and it
recombines to
the target site.
Safe harbor knockin:
Gene knockin at Safe harbors such as hROSA26 and AAVS1 not only
avoids
random insertion in genome, but also achieves overexpression of
target
gene.
Case Study:
The most common method to treat hemophilia is substitution therapy,
but
this method has the risk of virus infection, and it is a method that
needs lifelong continuous treatment. Gene therapy seems like the
only
way can cure hemophilia. CRISPR/Cas9 technology can be used for gene
therapy of hemophilia. The mutations of coagulation factors, F8 and
F9,
are the main causes of hemophilia. Previous studies have shown that
F9
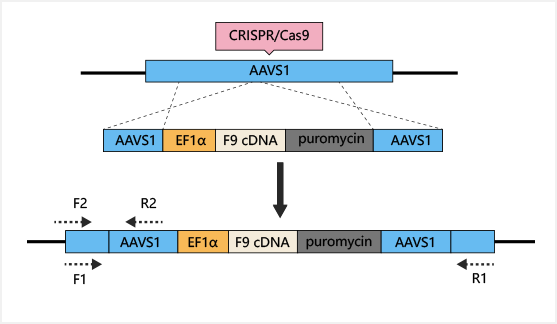
is a more effective gene therapy target. AAVS1-Cas9-sgRNA plasmid
and
AAVS1-EF1α-F9 cDNA puromycin donor plasmid were constructed and
transferred into iPSC. Human factor IX (hFIX) antigen activity was
detected in the culture supernatant. Finally, liver cells
differentiated
from iPSC were transplanted into NOD/SCID mice by spleen injection,
to
cure hemophilia B.
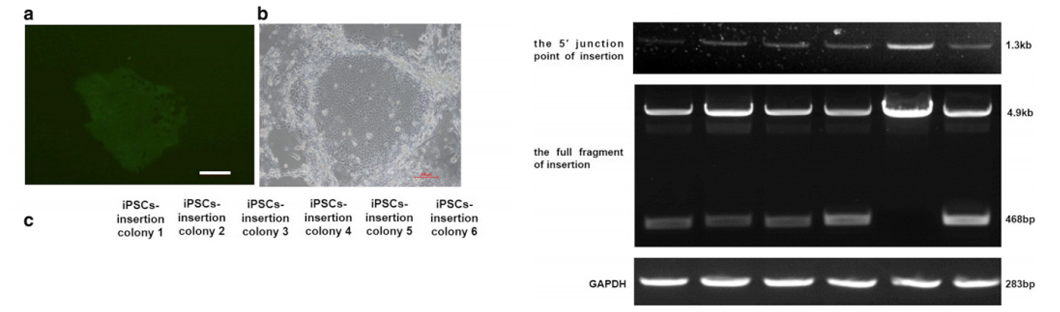
After 48 hours of transfection, puromycin was used for drug
screening.
Most iPSCs died after drug screening, but a few survived. After
about 7
days, each surviving iPSC clone was expended to be further testing
of
insertion (Fig. a, b). Six clones were selected. As shown in Figure
C,
1.3kb fragments can be detected in all iPSC clones with primers;
468bp
and 4.9kb fragments can be detected in iPSC clones 1, 2, 3, 4 and 6
with
another pair of primers, indicating F9 cDNA heterozygous insertion;
only
4.9kb fragments can be detected in iPSC clone 5, indicating F9 cDNA
homozygous insertion.
Reference:
Lyu, Cuicui, et al. "Targeted genome engineering in human induced
pluripotent stem cells from patients with hemophilia B using the
CRISPR-Cas9 system." Stem cell research & therapy 9.1 (2018): 92.















 Phone
Phone
 Contact
Contact

